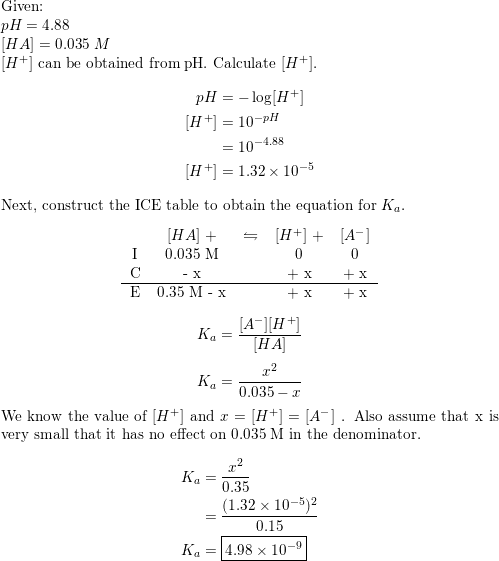Chapter 16C - Calculating Ka from the pH The pH of a 0.10 M solution of formic acid HCOOH at 25 C is 2.38. Calculate Ka for formic acid at | Course Hero

Acid-Base Buffers Equation & Examples | How to Calculate pH of a Buffer - Video & Lesson Transcript | Study.com










![Solved 3. Calculate the [H3O+], [OH-], pH and pOH of a 0.120 | Chegg.com Solved 3. Calculate the [H3O+], [OH-], pH and pOH of a 0.120 | Chegg.com](https://media.cheggcdn.com/study/f5c/f5c5a593-319b-4d2b-9ea3-2e5d84e0d782/image)
![Calculating [H+] and pH from Ka Calculating [H+] and pH from Ka](https://www.mi.mun.ca/users/pfisher/chemistry1011_134/img013.gif)