![Calculate from the Van der Waals equation, the temperature at which 192g of SO2 would occupy a volume of 6 dm^3 at 15 atm pressure [a = 5.68 atm L^2 mol^-2, b = 0.06 L mol^-1] Calculate from the Van der Waals equation, the temperature at which 192g of SO2 would occupy a volume of 6 dm^3 at 15 atm pressure [a = 5.68 atm L^2 mol^-2, b = 0.06 L mol^-1]](https://dwes9vv9u0550.cloudfront.net/images/2086003/05177f32-83c6-4e02-85a6-3b11431b23aa.jpg)
Calculate from the Van der Waals equation, the temperature at which 192g of SO2 would occupy a volume of 6 dm^3 at 15 atm pressure [a = 5.68 atm L^2 mol^-2, b = 0.06 L mol^-1]

A balloon filled with helium gas at 1.00 atm occupies 11.1 L. What volume would the balloon occupy in the upper atmosphere, at a pressure of 0.25 atm and a constant temperature?

SOLVED: If a balloon holds 3.7 L of gas at 25 oC, what will be the volume of the balloon at 46 oC?Assuming constant pressure.

A balloon is filled to a volume of 2.20L at a temperature of 22°C . The ballon is then heated to temperature - Brainly.ph
What would be the volume of the balloon that at sea level has a volume of 5 liters when the pressure will drop to p2=0.25 ATM? - Quora

On heating a flask, at constant pressure, from 27% to 227^oC,200 mL of air was expelled out .what was the volume of the flask?

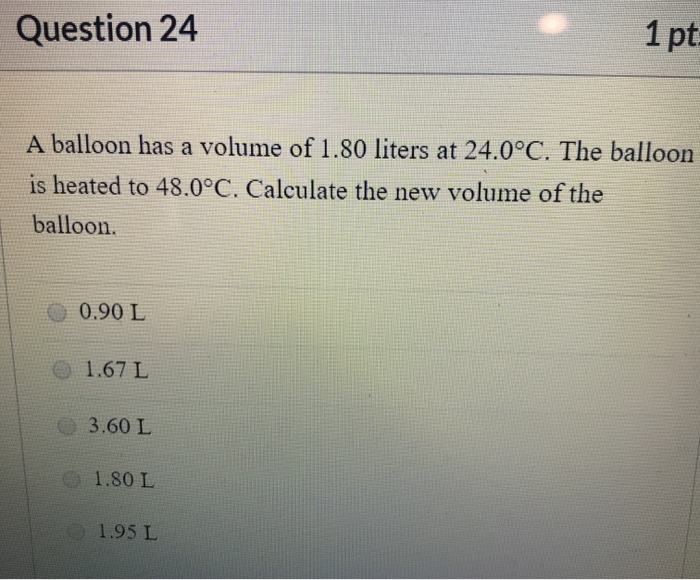
SOLVED: A balloon has a volume of 1.80 liters at 24.0*C. The balloon is heated to 48.0*C. Calculate the new volume of the balloon a. 1.95 L b. 1.80 L c. 1.67